Building Momentum Towards a Cure
One of the major challenges within a foundation supporting medical research and patients is to determine priorities. FSR has been extremely strategic in determining how our organization can best lead to better lives for patients. One of our primary priorities is to accelerate sarcoidosis research which will lead to treatments and possibly a cure.
Our strategy is to leverage our capacity to ensure other stakeholders push their resources into the sarcoidosis disease space. One of the main objectives of FSR is to “prime the pump” within the sarcoid research space – to make the field of sarcoidosis research more inviting to those with much more extensive resources by filling some of the current gaps that serve as barriers (i.e. lack of data, lack of funding, lack of investigators, lack of awareness, lack of knowledge, etc.)
If FSR creates basic tools to help bridge these gaps, then bigger players, with much deeper pockets, will be enticed to join the field. We are literally building a case for why sarcoidosis is a viable field for industry, government and academia to enter. We build this case by addressing some of the basics gaps that currently keep them away.
FSR has developed a specific Scientific Research Agenda that outlines the main areas in which FSR will invest our resources. This was developed by our Scientific Advisory Board by identifying what “gaps” exist in sarcoidosis research where, if filled, could result in the maximum impact. We recognize our role is to bridge these gaps in various ways, including direct funding of studies, initiatives, and trials.
FSR invests our resources into 4 main research areas:
- Disease Model – FSR has made history in the sarcoidosis space by awarding a total of $750,000 to FIVE Grant Awardees in grant funding toward the development of a sarcoidosis disease model. The lack of a disease model creates a major barrier to additional research in the space – once researchers have multiple sound models they can utilize within the sarcoidosis research, pipelines to increased research will open. The FSR grants, $150,000 per awardee, were provided via a competitive grant process to investigators conducting research toward this area. Research findings and information on the next phase of grant funding to further this work will be shared worldwide.
- Patient Registry – Currently over 5,000 patients are registered and in 2017, we are launching an engagement plan that will lead to thousands more joining. Three scientific papers are in development for 2018 publication overviewing the registry and the data collected to date, the in-depth data has been shared with multiple partners representing both academia and industry as FSR continues to resource investigators with patient outcomes to push research forward. As the registry seeks to track the changes within each patient’s journey with the disease, registry participants will soon be invited to update their ongoing information to show how the disease is affecting them and how their treatments, medical outcomes, and challenges are changing.
- End Point Development – In helping dire determination and consensus around what is the ideal measurements for assessing outcomes in trials and other treatments, FSR is in development of Endpoint Committee and culminating Summit. This project will include participation of all stakeholders, including patients, clinicians, investigators, pharma, government, and other experts to work together in ongoing groups to develop guidelines that can be proposed to professional medical groups to guide trials and treatments. Launched in 2017 as lead by project chairs, this project will culminate with a large Summit for final guidelines development.
- Clinical Studies Network –Launched in 2015, the 8-member consortium has increased membership to 12 sites and brings together an international partnership of world-renowned medical institutes and researchers for unprecedented collaboration in studies and drug trials toward the treatment of sarcoidosis. The first study measuring patient outcomes will be presented in a published report which will help move the needle forward in sarcoidosis research. In addition, FSR is partnering with pharma industry and others to utilize the CSN for additional, concurrent studies that can deliver valuable information. The second CSN grant period includes increased grant funding to a total of $800,000 provided to sites. The FSR-CSN will continue to conduct studies addressing unanswered questions in sarcoidosis research alongside its partners in industry and academia.
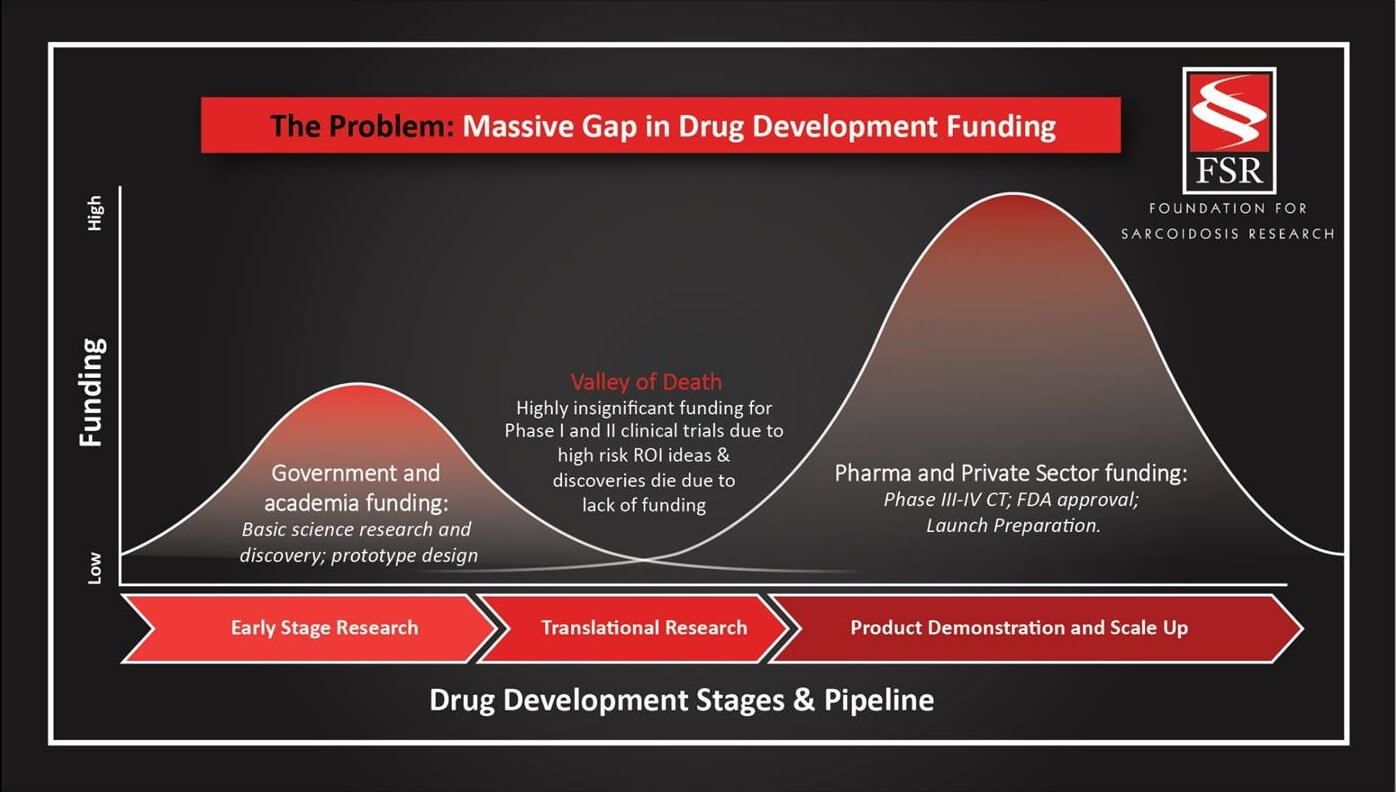
FSR embraces a model of medical research support that counters the traditional establishment. We seek to support, through direct funding or grant awards, projects that may be high risk and that have very little funding prospects from industry and government.
The current research system focuses on low-risk, low-yield to initiate reliable projects and studies. The approach has typically shown extremely slow advancement rates with little linking the research to actual patient therapies. FSR applies strategic use of our capital and collaboration to address the common “valley of death” need and to support more high-risk, innovative research.
Bridging the Valley of Death in Drug Development
Meet our thought leaders, including our Board of Directors and Scientific Advisory Board.